|
Ten of the 98 elements only exist in trace amounts. 
Eighty of the natural elements are stable, meaning that they cannot be subjected to radioactive decay. Thirty-two of the 98 elements are in their pure form. The first 98 elements listed in the periodic table occur naturally while the rest can only be found in nuclear accelerators and laboratories. 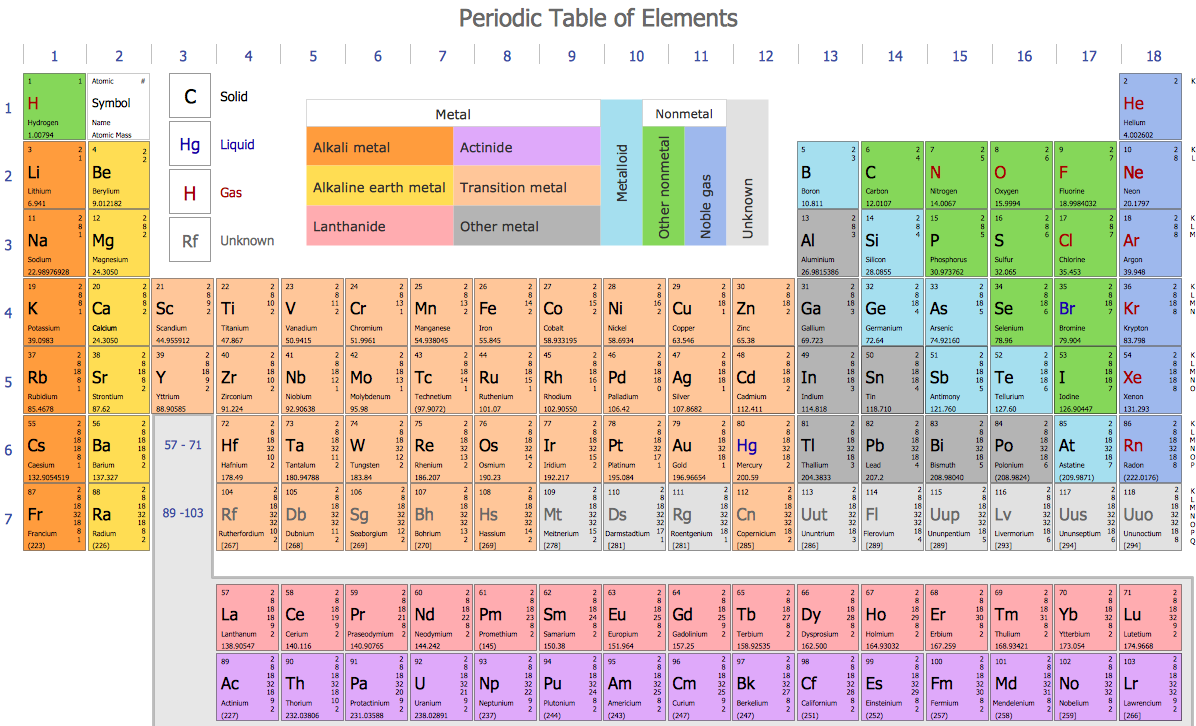
These are Nihonium (113), Moskovi (115), Tennessine (117), and Oganesson (118). Four of these were included on the list in 2016. The periodic table contains a total of 118 elements. Elements like copper, carbon, and silver have been in existence for thousands of years. Some elements like hydrogen and sodium are popular while others like dysprosium remain unknown because they are rarely used. While the elements on the right side of the periodic table are non-metals. The elements found on the left side of the periodic table are typically metals. Elements can be categorized into three major groups that include metals, nonmetals, and metalloids. The atomic number refers to the number of protons found in the atom of an element. The periodic table outlines each element’s electron configuration, the atomic number of the element, and the chemical properties of the element. Properties of ElementsĮvery known element has a name and a number, which are listed in the periodic table. When the number of protons in an atom is changed, then the element changes. The atoms in an element contain the same number of protons, but the number of neutrons varies. However, they can only be transformed into other elements by nuclear procedures. Elements cannot be broken further into smaller substances using chemical reactions. All the atoms in a particular element bear the same atomic number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
